The world has been facing the COVID-19 global pandemic for several months now, yet new symptoms are still being discovered. According to a recent study, 45% of patients with severe manifestations of COVID-19 experience neurological damage and neurological symptoms that may – in the long-term – develop into depression, insomnia, Parkinson’s disease, memory loss, or accelerated brain aging processes (1,2). The scientific community today mentions the urgent need to understand the neurological manifestations of COVID-19, starting with the systematic analysis of brain MRI findings and the neurological follow-up of patients (3,4).
Qynapse is developing collaborations with European and US hospitals to support the diagnosis and monitoring of neurological complications of the virus, by providing QyScore® to automatically quantify brain MRI markers in COVID-19 patients for retrospective or prospective analyses. Since the literature highlights the concept that the invasion of the central nervous system by SARS-CoV-2 may occur through the olfactory system (5,6), one of the first collaborations initiated with the University Hospital of Verona in Italy will investigate the long-term brain damages in COVID-19 patients presenting the frequently found minor symptoms of acute loss of smell (anosmia) and taste (ageusia) (7).
According to Dr. Maria Paola Cecchini, from the Department of Neurosciences, Biomedicine and Movement Sciences, Anatomy and Histology Section of the University of Verona: “This important work will support a better understanding of the post-infectious brain damages and the correlation between neuroimaging data and chemosensory function.“ As further highlighted by Dr. Francesca Pizzini, from the Department of Diagnostics and Public Health, Radiology Section of the University of Verona: “This study will be crucial to better characterize the underlying mechanisms of the virus on the central nervous system.”
With this collaboration, Qynapse wishes to share its knowledge and intellectual efforts to contribute to improving the comprehension of COVID-19 neurological manifestations for better patient care.
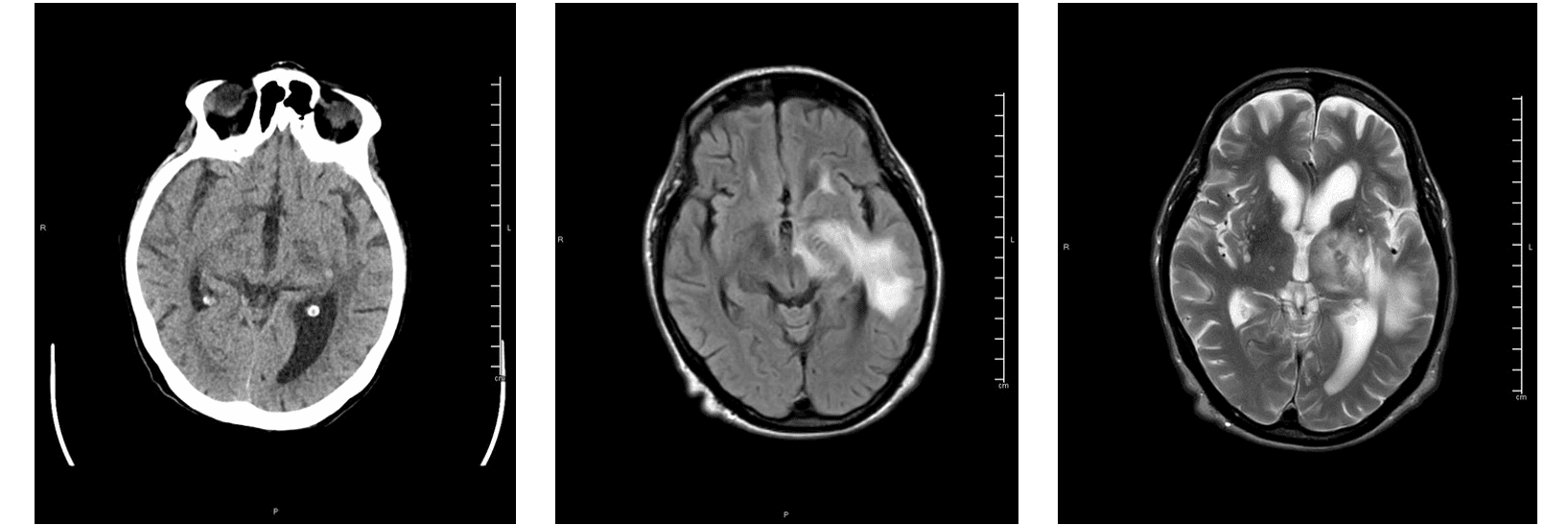
CT and MRI images showing a lesion centered at left thalamus, the internal capsule, basal ganglia and extending to the temporal white matter with evidence of hemorrhage indicated by hyperdense focus at CT. These findings are in line with the Acute Hemorrhagic Necrotizing Encephalopathy found in a COVID-19 positive Patient. (Images provided by Pizzini and Beltramello,University of Verona).
About QyScore® : QyScore® allows automated quantification of a broad portfolio of imaging markers crucial for the early diagnosis and clinical monitoring of brain disorders, in the context of clinical trials or clinical routine. The software supports clinicians by providing highly reproducible results to eliminate the intra/inter-rater variability in image reading and reduce the time and costs associated. QyScore® is an FDA-cleared (Class II) and CE-marked (Class IIa) medical device software.
Contact Qynapse contact@qynapse.com to discuss potential collaborations.
References: 1. Mao L, Jin H, Wang M, et al., JAMA Neurol. 2020;77(6):683–690. 2. Fotuhi M, et al., Journal of Alzheimer’s Disease, 2020 3. The neurological impact of COVID-19. Lancet Neurol. 2020 4. Kremer S, Lersy F, de Sèze J, et al. Radiology 2020 5. Politi LS, Salsano E, Grimaldi M. JAMA Neurology 2020 6. DosSantos MF, Devalle S, Aran V, et al. Front Neuroanat. 2020 7. Lee Y, Min P, Lee S, Kim SW. J Korean Med Sci. 2020